 These studies demonstrated that olmesartan-based antihypertensive strategy may indeed provide sustained BP control over the 24-h period in a wide proportion of hypertensive patients, thus contributing to a substantial progress in hypertension management. After a brief description of the pharmacological characteristics of olmesartan, we will provide a thorough overview of the clinical studies that investigated its efficacy and safety in the clinical management of hypertensive patients both in monotherapy and in dual combination therapies with either thiazide diuretics or calcium channel blockers. These characteristics produce effective and sustained blood pressure reductions in hypertensive patients at different cardiovascular risk profile with a good tolerability profile. Olmesartan medoxomil is an antihypertensive drug of the class of angiotensin II type 1 (AT1) receptor antagonists (or blockers), characterized by tight and prolonged binding to AT1 receptor compared to other molecules within the same class. The wider use of combinations, especially in fixed doses, such as the calcium channel blocker amlodipine, and the angiotensin II receptor antagonist olmesartan for the treatment of hypertension, will help improve blood pressure monitoring, reduce the risk of complications and increase the AH patients’ life expectancy. The need for not only wider combination therapy for the treatment of hypertensive patients, but also its earlier commencement has been strongly confirmed by these new guidelines for the management of AH. This is due, among other things, to changing the blood pressure levels classification and categorizing all adults with blood pressure of 130/80 mm Hg. The new US guidelines for the management of hypertension indicate that the target blood pressure level for all patients was less than 130/80 mm Hg. The key goal in the treatment for patients with AH is to reduce the risk of cardiovascular complications through reaching the target level of arterial pressure (AP) and organ protection, treatment of concomitant diseases and correction of risk factors. An AML/OM-based titration regimen effectively reduces BP in patients with hypertension. The study drug was well tolerated with few adverse events (peripheral edema, 2.2% dizziness, 1.1%). Cumulatively, 76.8% of patients uptitrated to AML/OM 10/40 mg/day attained an SeBP goal of <140/90 mmHg. Proportions of patients achieving mean 24-hour ambulatory BP prespecified study targets were 70.9% (<130/80 mmHg), 48.3% (<125/75 mmHg), and 40.7% (<120/80 mmHg). At baseline, the mean SeBP (± SD) was 158.2 ± 12.6/92.8 ± 8.6 mmHg and at week 12, the mean SeBP change (± SEM) from baseline (last observation carried forward) was -24.1 ± 1.1/-12.1 ± 0.7 mmHg (p < 0.0001 versus baseline). 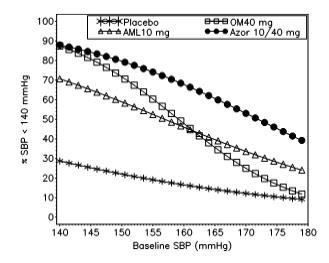
At week 12, the change from baseline in mean 24-hour ambulatory BP (± standard error of the mean ) was -21.4 ± 0.8/-12.7 ± 0.5 mmHg (p < 0.0001 versus baseline). 
At baseline, the mean 24-hour ambulatory BP (± standard deviation ) was 144.8 ± 11.1/85.7 ± 7.9 mmHg. The primary efficacy endpoint was the change from baseline in mean 24-hour systolic BP at week 12 as assessed by ABPM. Patients were initially treated with AML 5 mg/ day and uptitrated to AML/OM 5/20, 5/40, and 10/40 mg/day every 3 weeks if mean seated BP (SeBP) was ≥ 120/80 mmHg. 
This 12-week, titrate-to-goal study was conducted in 185 patients with hypertension. The aim of the present study was to use ambulatory blood pressure (BP) monitoring (ABPM) to determine the efficacy of a fixed-dose combination of amlodipine (AML) and olmesartan medoxomil (OM) over the 24-hour dosing interval. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
